Leave Your Message
Cationic surfactants are gaining attention for their cleaning efficacy. Recent studies show they can enhance soil removal by up to 30%. This is crucial for various industries, including healthcare and personal care. According to industry expert Dr. Emily Carter, “Cationic surfactants offer unique properties that can revolutionize cleaning processes.”
Many products already incorporate these surfactants due to their antibacterial and antistatic properties. However, challenges remain. Some formulations can lead to residue build-up, which affects performance. Achieving the right concentration for optimal results is not always straightforward.
The market is evolving rapidly, driven by consumer demand for eco-friendly options. It's essential to balance performance with sustainability. Continuous research is needed to refine formulations. The goal is to create cationic surfactants that deliver maximum cleaning power with minimal environmental impact.

Cationic surfactants play a crucial role in cleaning applications. They possess unique properties that enhance cleaning performance. These surfactants have a positive charge, allowing them to bind effectively to negatively charged surfaces. This feature improves their ability to remove dirt and bacteria. Often, they work well in fabric softeners and shampoos.
The solubility of cationic surfactants in water is noteworthy. They tend to create a rich lather, which helps in spreading evenly during cleaning. However, achieving the right concentration is key. Too much can lead to residue build-up, negatively impacting the cleaning process. In some cases, users may experience irritation on sensitive skin. Understanding the right balance is essential.
Another important aspect is their antibacterial properties. Cationic surfactants can disrupt bacterial cell membranes, providing additional cleaning effectiveness. Yet, the environmental impact of these substances raises concerns. Some formulations may not degrade easily, posing a risk to aquatic life. As we strive for improved cleaning performance, a critical evaluation of their use is necessary.
Cationic surfactants play a significant role in enhancing cleaning performance. They possess a positive charge, which allows them to effectively interact with negatively charged dirt and stains. This unique property creates a strong affinity for various surfaces, making them ideal for applications in detergents and fabrics.
There are several types of cationic surfactants. Quaternary ammonium compounds, or quats, are widely used due to their antimicrobial properties. They not only clean but also disinfect surfaces. However, their effectiveness can vary based on concentration and the presence of organic matter. Some users report that quats can leave a residue, which sometimes raises concerns.
Another type includes fatty amines, known for their ability to emulsify and stabilize formulations. While they are efficient, their environmental impact is a consideration. Users must weigh the benefits against potential ecological effects. It’s essential to experiment and adapt formulations based on specific cleaning needs. Finding the right balance can be challenging, but it's crucial for optimal cleaning results.
Cationic surfactants play a critical role in cleaning formulations. They possess a positive charge, which allows them to interact effectively with negatively charged surfaces. This interaction enhances the removal of dirt and bacteria, making cleaning more efficient.
According to a recent industry report by the Surfactant Association, cationic surfactants can increase cleaning performance by up to 40%. Their effectiveness is particularly notable in fabric softeners and disinfectants. The mechanism involves the surfactants binding to dirt particles, facilitating their removal during rinsing. This is particularly evident in textile applications, where they can improve fabric feel and resilience.
However, not all cationic surfactants are created equal. Some may leave residue after cleaning, counteracting their benefits. In personal care products, they can cause skin irritation for certain users. Adjusting concentrations is essential. Overuse can lead to suboptimal cleaning and wasted resources. Balancing efficacy and safety remains a challenge for formulators in the industry.
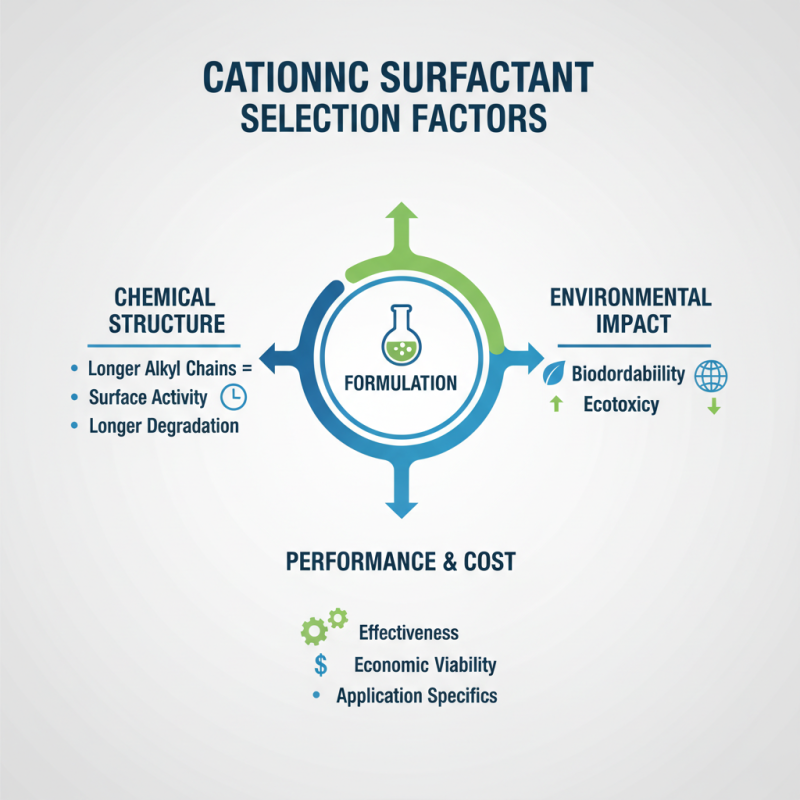
When selecting cationic surfactants for formulations, various factors come into play. The effectiveness of these surfactants depends significantly on their chemical structure. Longer alkyl chains often enhance the surfactants' surface activity. However, this can also lead to longer degradation times. Environmental impact is a growing concern.
Another important factor is the target application. Cationic surfactants function differently in personal care, cleaning, and industrial applications. For instance, in hair conditioners, they help provide softness and manageability. Yet, they can create buildup on hair over time. This raises questions about balance in formulation.
Finally, the regulatory landscape affects choices as well. Compliance to safety standards is crucial, which sometimes constrains formulation options. This can lead to challenging scenarios where performance may be sacrificed for safety. Manufacturers must navigate these complexities thoughtfully.
As the cleaning industry evolves, cationic surfactants are gaining attention. These compounds excel in their ability to clean surfaces effectively. Yet, there are gaps in research regarding their long-term impacts on health and the environment.
Sustainable solutions are becoming essential. Many cationic surfactants degrade slowly, leading to potential accumulation in ecosystems. The challenge lies in creating products that are both effective and biodegradable. Alternatives are needed. Research into plant-derived surfactants is promising, but practical applications remain limited.
Innovation is vital. New formulations must focus on reducing chemical footprints. It is vital to explore natural sources that are safe and efficient. There's a need for better collaboration among scientists, manufacturers, and regulators. Experimentation with new materials could lead to breakthroughs. Balancing performance with sustainability is complex. The future of cationic surfactants may lie in reformulated, eco-friendly solutions.